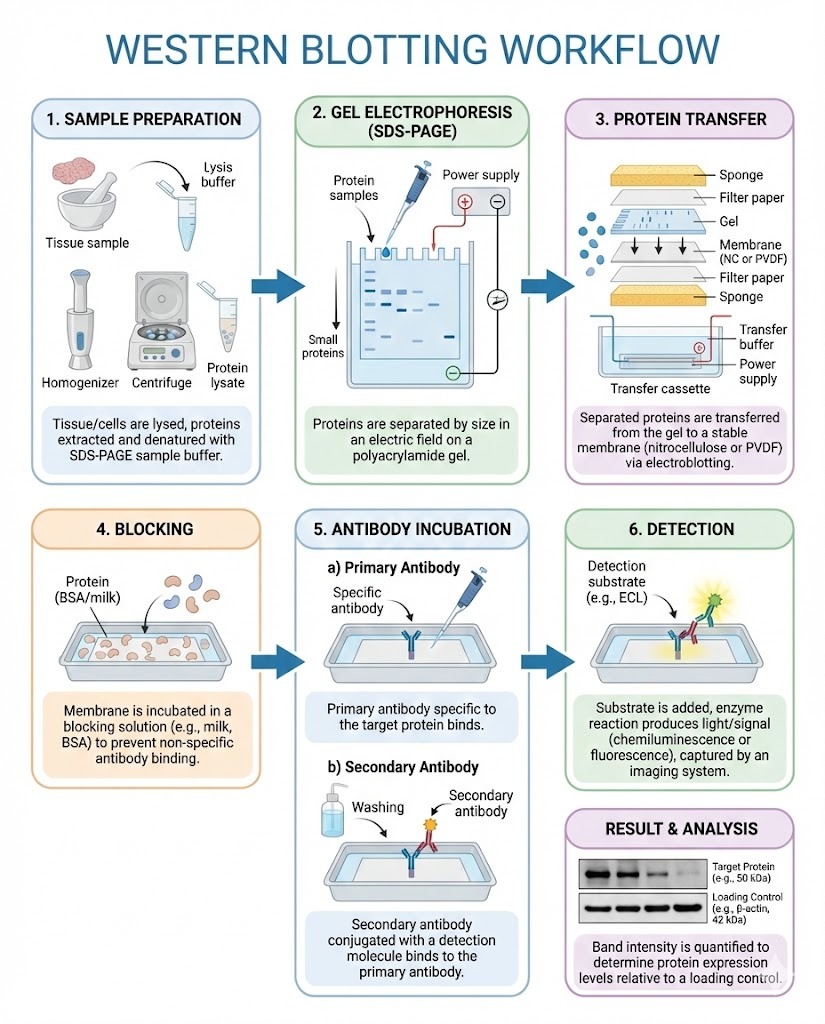
Protein detection and quantification remain central to biological and biomedical research. Among available techniques, western blotting continues to be one of the most widely used due to its specificity, flexibility, and relatively low cost. At the heart of successful experiments is a well-structured Western Blotting Workflow, which defines how samples are prepared, separated, transferred, detected, and analyzed.
Understanding each stage of this workflow helps laboratories generate reproducible results, reduce experimental errors, and improve overall data quality. This guide explains the full process in practical terms and highlights best practices used in research and diagnostic environments.
What Is the Western Blotting Workflow?
The Western Blotting Workflow refers to the complete sequence of steps used to detect specific proteins in complex biological samples. It begins with sample preparation and ends with data interpretation.
Each stage influences the next. Inconsistent handling early in the process can compromise signal strength, specificity, or quantification later on. A standardized workflow ensures that results are accurate, comparable between experiments, and suitable for publication or regulatory review.
Why Workflow Design Matters
Western blotting involves multiple manual steps, reagents, and instruments. Without a consistent workflow, laboratories often encounter:
-
High background noise
-
Weak or inconsistent bands
-
Poor reproducibility
-
Misinterpretation of protein expression levels
A well-defined Western Blotting Workflow helps minimize technical variability and supports long-term project reliability.
Core Stages of the Western Blotting Workflow
Although protocols vary by application, most workflows follow the same fundamental structure.
1. Sample Collection and Protein Extraction
The process begins with biological samples such as cells, tissues, or bodily fluids. Proteins are extracted using lysis buffers that break down cell membranes while preserving protein integrity.
Key considerations include:
-
Choosing appropriate lysis buffers
-
Maintaining cold temperatures to prevent degradation
-
Adding protease and phosphatase inhibitors
-
Measuring total protein concentration
Accurate protein quantification ensures equal loading across gel lanes.
2. Sample Preparation and Denaturation
Before separation, protein samples are mixed with loading buffer and heated. This step:
-
Denatures proteins
-
Applies a uniform negative charge
-
Breaks disulfide bonds
These actions allow proteins to separate primarily based on molecular weight during electrophoresis.
3. Gel Electrophoresis
Proteins are loaded onto a polyacrylamide gel and separated by size using an electric field.
Important factors include:
-
Gel percentage selection
-
Voltage and run time
-
Sample volume consistency
Smaller proteins migrate faster, while larger proteins move more slowly through the gel matrix.
4. Transfer to Membrane
After separation, proteins are transferred from the gel to a membrane, usually nitrocellulose or PVDF.
Transfer methods include:
-
Wet transfer
-
Semi-dry transfer
-
Dry transfer systems
Efficient transfer is critical, as incomplete or uneven transfer can lead to missing or distorted bands.
5. Membrane Blocking
Blocking prevents nonspecific antibody binding by covering unoccupied sites on the membrane.
Common blocking agents:
-
Nonfat dry milk
-
Bovine serum albumin
-
Commercial blocking buffers
Insufficient blocking increases background noise, while excessive blocking may reduce signal intensity.
6. Antibody Incubation
This stage defines the specificity of the Western Blotting Workflow.
It involves:
-
Incubation with a primary antibody specific to the target protein
-
Washing to remove unbound antibodies
-
Incubation with a labeled secondary antibody
The secondary antibody is typically conjugated to an enzyme or fluorescent tag for detection.
7. Detection
Detection methods depend on the labeling system used.
Common approaches:
-
Chemiluminescence
-
Fluorescence
-
Colorimetric detection
Modern imaging systems capture and quantify signal intensity, producing digital data for analysis.
8. Data Analysis and Interpretation
Final steps include:
-
Measuring band intensity
-
Normalizing to housekeeping proteins
-
Comparing experimental groups
-
Statistical evaluation
Proper documentation ensures that results can be reviewed and reproduced.
Factors That Influence Workflow Performance
Several variables affect the success of western blotting.
Key factors include:
-
Antibody specificity and quality
-
Protein loading accuracy
-
Transfer efficiency
-
Blocking conditions
-
Washing stringency
-
Detection sensitivity
Optimizing these variables strengthens the overall Western Blotting Workflow.
Common Applications
Western blotting remains widely used across research disciplines.
Typical applications include:
-
Confirming gene expression at the protein level
-
Studying post-translational modifications
-
Validating antibody specificity
-
Investigating signaling pathways
-
Supporting diagnostic research
Its versatility allows integration into genomics, proteomics, pharmacology, and clinical studies.
Challenges and Limitations
Despite its popularity, western blotting has known limitations.
-
Labor intensive
-
Semi-quantitative unless carefully controlled
-
Sensitive to experimental variation
-
Limited throughput compared to automated platforms
Understanding these constraints helps researchers design realistic experiments and avoid overinterpretation.
Improving Efficiency Through Automation
Many laboratories now incorporate semi-automated or fully automated systems to improve consistency.
Benefits of automation:
-
Reduced hands-on time
-
Standardized reagent handling
-
Lower error rates
-
Improved reproducibility
While automation increases upfront cost, it often reduces long-term labor and repeat experiments.
Best Practices for a Reliable Western Blotting Workflow
Experienced laboratories follow several core principles.
-
Use validated antibodies from reputable suppliers
-
Include positive and negative controls
-
Maintain detailed protocols
-
Calibrate equipment regularly
-
Perform replicate experiments
-
Document reagent lot numbers and conditions
These practices improve confidence in experimental outcomes.
Choosing Reagents and Equipment
Reliable results depend heavily on reagent quality and instrumentation.
Selection criteria often include:
-
Compatibility with sample type
-
Sensitivity requirements
-
Budget constraints
-
Technical support availability
-
Long-term supply consistency
Many suppliers offer integrated western blotting kits that simplify workflow design and reduce troubleshooting.
Conclusion
The Western Blotting Workflow is a foundational process in protein research, transforming raw biological samples into interpretable molecular data. Each step, from protein extraction to signal detection, contributes to the accuracy and reproducibility of the final result.
By standardizing procedures, selecting high-quality reagents, and implementing best practices, laboratories can achieve consistent performance and reliable insights into protein expression and function. Whether used in academic research, biotechnology development, or diagnostic validation, a well-designed workflow remains essential for dependable western blotting outcomes.
Frequently Asked Questions
What are the main steps in the western blotting workflow?
The main steps include protein extraction, sample preparation, gel electrophoresis, transfer to membrane, blocking, antibody incubation, detection, and data analysis.
How long does a typical western blotting workflow take?
Depending on protocol complexity, the process can take from one day to several days, especially when including overnight antibody incubations.
Why is blocking important in western blotting procedures?
Blocking prevents nonspecific antibody binding, reducing background noise and improving signal clarity during detection.
How can reproducibility be improved in a western blotting workflow?
Reproducibility improves through standardized protocols, validated antibodies, consistent sample preparation, proper controls, and detailed documentation of experimental conditions.