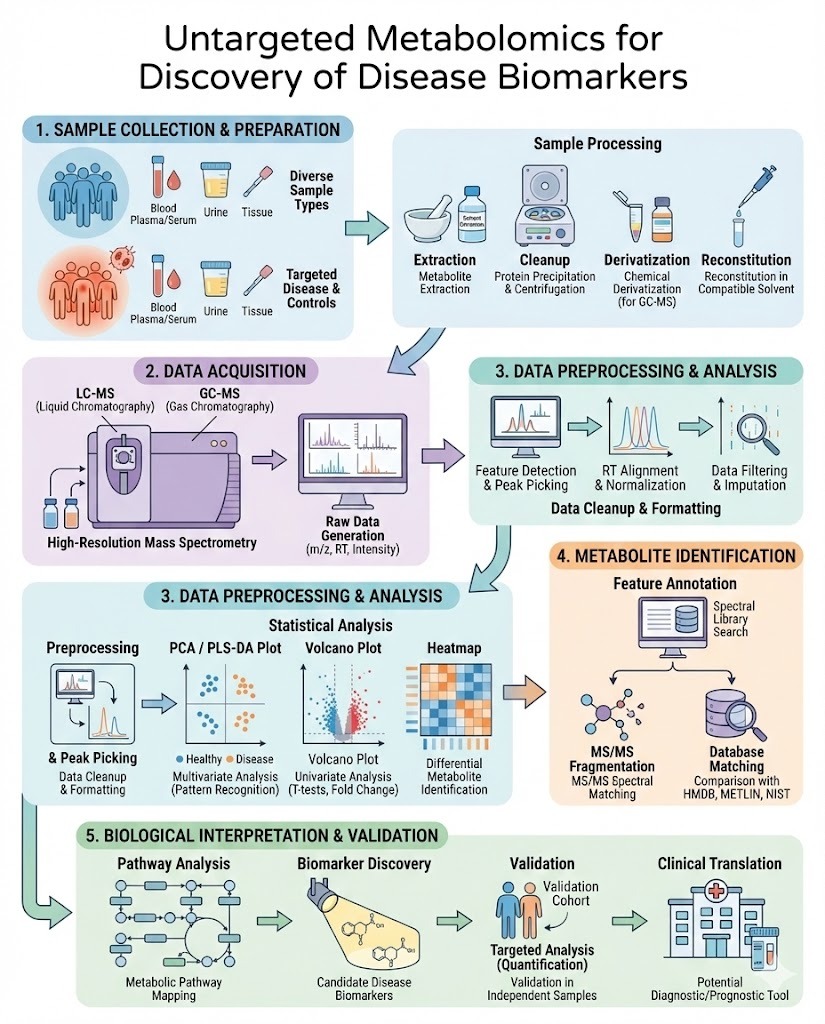
Untargeted metabolomics for discovery of disease biomarkers has become a cornerstone approach in modern biomedical research. By enabling comprehensive profiling of small molecules in biological systems, this methodology provides a powerful way to uncover biochemical changes associated with disease onset, progression, and therapeutic response.
Unlike targeted metabolomics, which focuses on predefined compounds, untargeted metabolomics surveys thousands of metabolites simultaneously without prior assumptions. This unbiased strategy is particularly valuable for identifying novel biomarkers, understanding disease mechanisms, and supporting precision medicine initiatives.
This article explores how untargeted metabolomics works, why it is uniquely suited for biomarker discovery, and how it is applied across clinical and translational research.
What Is Untargeted Metabolomics
Untargeted metabolomics is an analytical approach designed to measure as many metabolites as possible in a biological sample such as blood, urine, cerebrospinal fluid, or tissue extracts.
Key characteristics include:
-
Broad coverage of known and unknown metabolites
-
No predefined metabolite list
-
High resolution analytical platforms
-
Data driven hypothesis generation
The approach captures metabolic fingerprints that reflect genetic, environmental, dietary, and disease related influences.
Why Metabolomics Is Ideal for Biomarker Discovery
Metabolites represent the end products of cellular processes. Changes in their concentrations often occur earlier than clinical symptoms or structural tissue damage, making them ideal candidates for early disease detection.
Untargeted metabolomics for discovery of disease biomarkers offers several advantages:
-
Detects subtle biochemical changes
-
Reflects real time physiological status
-
Integrates signals from multiple biological pathways
-
Enables discovery of unexpected disease associations
-
Supports stratification of patient subgroups
These features make it especially valuable in complex diseases where single genetic or protein markers are insufficient.
Core Workflow in Untargeted Metabolomics Studies
The biomarker discovery process follows a structured pipeline.
Sample Collection and Preparation
Careful standardization is critical to avoid technical bias.
Key considerations include:
-
Fasting state of subjects
-
Sample storage temperature
-
Freeze thaw cycles
-
Anticoagulant choice for plasma samples
-
Extraction solvent selection
Analytical Platforms
The most commonly used technologies are:
-
Liquid chromatography mass spectrometry
-
Gas chromatography mass spectrometry
-
Nuclear magnetic resonance spectroscopy
Each platform offers unique strengths in sensitivity, metabolite coverage, and reproducibility.
Data Acquisition
High resolution instruments generate complex datasets containing:
-
Mass to charge ratios
-
Retention times
-
Signal intensities
-
Spectral fragmentation patterns
These raw data form the basis for downstream analysis.
Data Processing and Statistical Analysis
Processing includes:
-
Peak detection
-
Alignment across samples
-
Normalization
-
Noise filtering
Statistical techniques such as principal component analysis and multivariate modeling identify metabolites that differ significantly between disease and control groups.
Metabolite Identification and Validation
Putative biomarkers are matched to databases and confirmed using:
-
Reference standards
-
Tandem mass spectrometry
-
Independent cohorts
Validation ensures biological relevance and reproducibility.
Applications Across Disease Areas
Untargeted metabolomics for discovery of disease biomarkers has demonstrated value in numerous fields.
Oncology
Cancer studies use metabolomic profiling to:
-
Detect early stage tumors
-
Distinguish tumor subtypes
-
Monitor treatment response
-
Identify metabolic vulnerabilities
Cardiovascular Disease
Researchers identify biomarkers linked to:
-
Atherosclerosis progression
-
Myocardial injury
-
Lipid metabolism dysregulation
-
Inflammatory states
Neurological Disorders
Metabolomics supports investigation of:
-
Alzheimer’s disease
-
Parkinson’s disease
-
Multiple sclerosis
-
Epilepsy
Altered neurotransmitter metabolism and energy pathways often provide diagnostic clues.
Metabolic Disorders
In diabetes and obesity research, untargeted metabolomics reveals:
-
Insulin resistance markers
-
Lipid species associated with metabolic syndrome
-
Amino acid perturbations
-
Gut microbiome related metabolites
Infectious Diseases
Pathogen induced metabolic shifts enable:
-
Early infection detection
-
Differentiation between viral and bacterial causes
-
Monitoring of immune response
Advantages of Untargeted Metabolomics in Biomarker Research
This approach offers several technical and scientific benefits.
-
Comprehensive coverage of biochemical pathways
-
Ability to detect unknown metabolites
-
Minimal prior assumptions
-
Strong sensitivity to disease related perturbations
-
Compatibility with multi omics integration
These strengths support discovery oriented research where the underlying biology may not be fully understood.
Challenges and Limitations
Despite its value, untargeted metabolomics also presents challenges.
Data Complexity
Large datasets require advanced bioinformatics tools and expertise.
Metabolite Identification Bottleneck
Many detected features remain unidentified due to incomplete reference databases.
Biological Variability
Diet, medication, age, sex, and microbiome composition influence metabolite levels.
Reproducibility
Differences in sample handling, instrumentation, and data processing pipelines can affect results.
Clinical Translation
Biomarkers must undergo extensive validation before regulatory approval and clinical adoption.
Addressing these limitations requires standardization, rigorous study design, and cross laboratory collaboration.
Best Practices for Biomarker Discovery Studies
To maximize success, researchers should consider:
-
Adequate sample size and statistical power
-
Careful matching of cases and controls
-
Strict quality control procedures
-
Use of independent validation cohorts
-
Integration with clinical metadata
-
Transparent reporting of analytical methods
Following these principles strengthens the reliability of discovered biomarkers.
Role in Precision Medicine
Untargeted metabolomics for discovery of disease biomarkers plays a central role in personalized healthcare strategies.
It enables:
-
Identification of patient specific metabolic profiles
-
Prediction of drug response
-
Detection of adverse effects
-
Optimization of treatment selection
-
Monitoring of disease recurrence
By moving beyond population averages, metabolomics supports tailored therapeutic approaches.
Future Directions
Technological advances continue to expand the impact of metabolomics.
Emerging trends include:
-
Improved mass spectrometry resolution
-
Automated compound identification
-
Integration with genomics and proteomics
-
Machine learning based pattern recognition
-
Real time metabolic monitoring
These developments will further enhance biomarker discovery and clinical implementation.
Conclusion
Untargeted metabolomics for discovery of disease biomarkers represents a transformative approach in biomedical research. By capturing comprehensive metabolic signatures without prior assumptions, it enables identification of novel biomarkers, reveals disease mechanisms, and supports precision medicine initiatives.
Although challenges remain in data interpretation and clinical translation, continued technological and methodological improvements are steadily overcoming these barriers. As a result, untargeted metabolomics is increasingly positioned as a foundational tool in early diagnosis, therapeutic monitoring, and personalized healthcare strategies.
Frequently Asked Questions
What is untargeted metabolomics for disease biomarker discovery
It is an analytical approach that measures a wide range of metabolites in biological samples to identify biochemical changes associated with specific diseases, without focusing on predefined compounds.
How does untargeted metabolomics differ from targeted metabolomics
Untargeted metabolomics captures thousands of metabolites simultaneously in a discovery driven manner, while targeted metabolomics measures a specific set of known compounds with higher quantitative precision.
Why is untargeted metabolomics useful for identifying new biomarkers
Because it does not rely on prior knowledge, it can reveal unexpected metabolic alterations and novel disease related molecules that would be missed using targeted methods.
What diseases benefit most from untargeted metabolomics biomarker research
It is widely used in cancer, cardiovascular disease, neurological disorders, metabolic conditions, autoimmune diseases, and infectious disease research due to its sensitivity to complex biochemical changes.
Download PDF
Enter your email address to unlock the full PDF download.
Generating PDF...