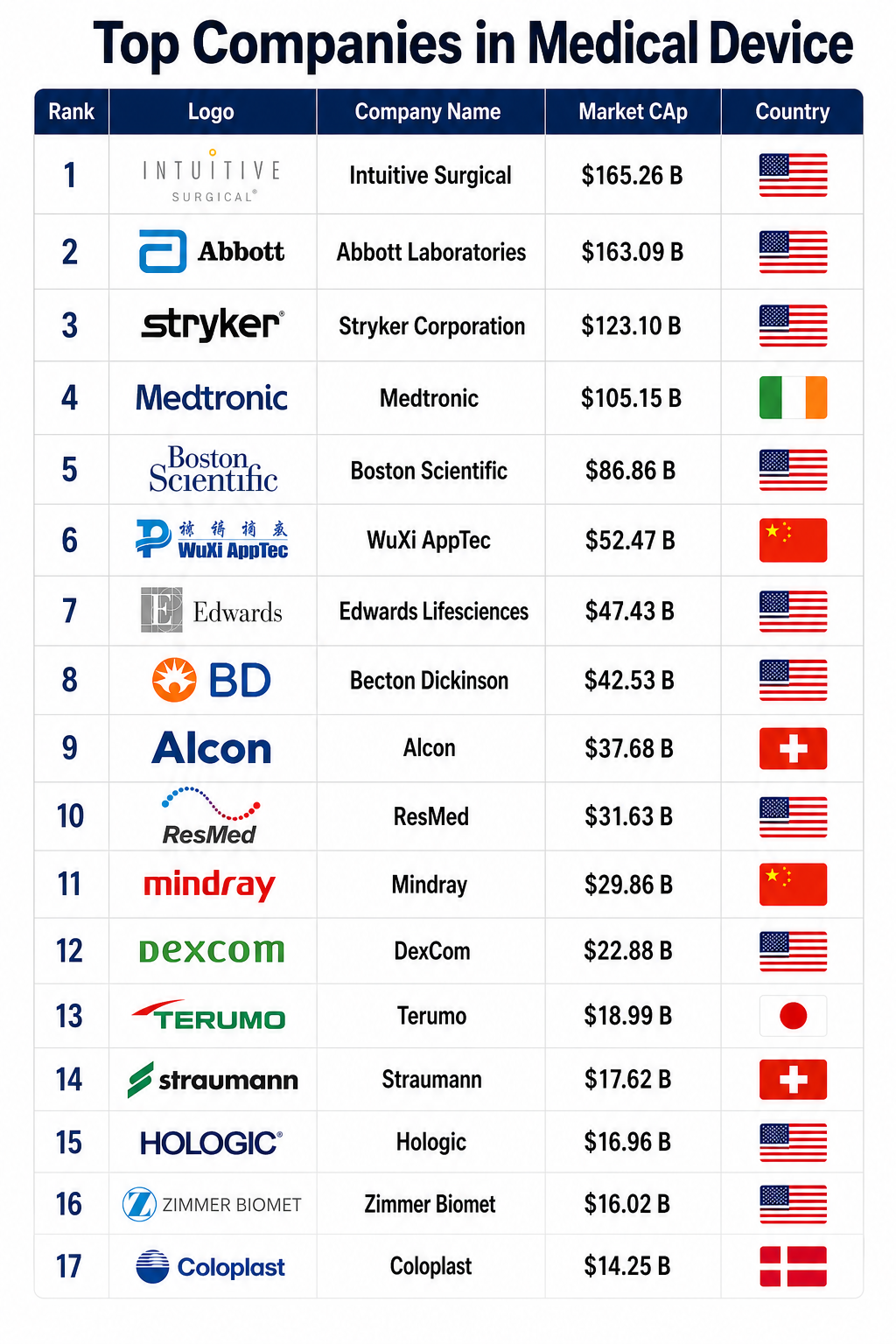
The medical device industry in 2026 stands at a historic crossroads, defined by the total integration of Artificial Intelligence (AI), a fundamental shift toward decentralized “home-based” care, and a regulatory landscape that has become more rigorous yet increasingly digitized. As of April 2026, the global medical devices market is valued at approximately $735 billion, driven by a compound annual growth rate (CAGR) of over 7%. This growth is not merely a reflection of increased sales but a byproduct of a paradigm shift from reactive treatment to predictive, data-driven wellness. The industry has moved beyond the “Internet of Medical Things” (IoMT) as a buzzword; it is now the operational backbone of global healthcare. Manufacturers are no longer just hardware providers but are now “Software as a Medical Device” (SaMD) developers, data custodians, and service partners.
The AI Revolution: From Tool to “Agentic” Partner
The most significant trend in 2026 is the evolution of AI within medical devices. We have moved past simple diagnostic algorithms to “Agentic AI”—systems capable of observing, planning, and acting autonomously under clinician oversight. AI-based devices now account for more than 33% of the technology segment. These systems are embedded in everything from imaging suites that flag anomalies in real-time to “smart” surgical instruments that provide haptic feedback to surgeons, preventing accidental tissue damage. For example, AI-enhanced endoscopy and robotic surgery platforms with compact, modular arms have become standard in both major hospitals and smaller ambulatory surgery centers (ASCs). These robots assist in high-precision procedures, significantly reducing surgeon fatigue and patient recovery times.
The Rise of Decentralized and “Hospital-at-Home” Tech
The home has become the new frontline for medical intervention. In 2026, the demand for Remote Patient Monitoring (RPM) and next-generation wearables has reached record highs. Devices that were once consumer-grade, like smartwatches, have evolved into medical-grade tools capable of continuous ECG monitoring, glucose sensing without skin pricks, and early detection of respiratory distress. We are seeing a surge in “Smart Implants”—orthopedic and cardiac implants that transmit data about inflammation or mechanical wear directly to a patient’s smartphone. Furthermore, portable dialysis machines and wearable artificial kidneys have begun to gain market traction, allowing patients with chronic renal failure to maintain their lifestyle without being tethered to a clinic three times a week.
Regulatory Tightening and the “Digital-First” Compliance
Navigating the 2026 regulatory environment requires unprecedented agility. The EU Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR) are in full effect, with the European database, EUDAMED, now serving as a mandatory, transparent ledger for every device on the market. Manufacturers must provide rigorous clinical evidence and maintain comprehensive post-market surveillance systems. In the United States, the FDA has moved toward a Total Product Life Cycle (TPLC) approach, aligning its Quality Management System Regulation (QMSR) with ISO 13485:2016. A key development this year is the widespread adoption of Predetermined Change Control Plans (PCCPs), which allow AI-enabled devices to update their algorithms without requiring a new regulatory submission for every iteration—provided the changes stay within pre-approved boundaries.
Supply Chain Resilience and Sustainability
The supply chain crises of the early 2020s have birthed a new model of “strategic resilience.” In 2026, medical device companies have moved away from “just-in-time” manufacturing toward “just-in-case” inventory management and nearshoring. Sustainability has also become a non-negotiable metric. Major distributors have transitioned to electric delivery fleets, and the industry is aggressively adopting circular economy principles—designing devices for easy disassembly, refurbishment, and recycling. 3D printing, or additive manufacturing, is now widely used for point-of-care production of custom prosthetics and surgical guides, reducing the carbon footprint of long-distance shipping and ensuring a perfect fit for the patient.
Market Geography and Future Outlook
While North America maintains the largest market share (approx. 40%), the Asia-Pacific region is the fastest-growing hub, fueled by massive infrastructure investments in China and India. By the end of 2026, the industry is expected to be more connected than ever. The focus for the remainder of the decade will be on interoperability—ensuring that a device made by one manufacturer can seamlessly share data with another’s platform. As we look toward 2027 and beyond, the medical device industry is no longer just about “fixing” the body; it is about providing a continuous, invisible safety net of data and technology that empowers patients and clinicians alike.