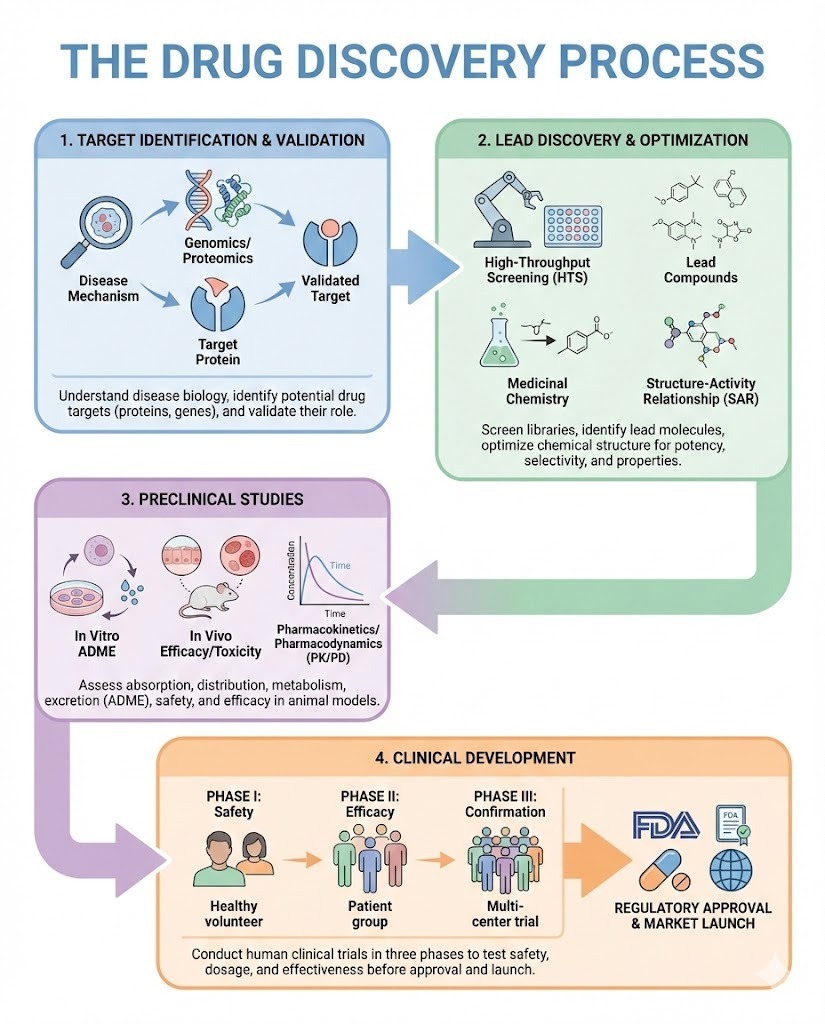
The development of new medicines is one of the most complex and resource-intensive efforts in modern science. At the heart of this effort is The Drug Discovery Process, a structured sequence of research activities designed to identify, validate, and optimize compounds that can safely and effectively treat disease.
This process combines biology, chemistry, data analysis, and clinical insight to transform basic scientific understanding into viable therapeutic candidates. For researchers, biotechnology companies, and pharmaceutical organizations, understanding how drug discovery works is essential for managing timelines, budgets, and scientific risk.
This guide provides a comprehensive overview of the major stages, key challenges, and best practices involved in the drug discovery lifecycle.
What Is the Drug Discovery Process?
The drug discovery process refers to the early and middle phases of pharmaceutical development that occur before formal clinical trials begin. It focuses on identifying a biological target, discovering compounds that interact with it, and refining those compounds into drug candidates suitable for testing in humans.
It is distinct from drug development, which includes clinical trials, regulatory approval, and commercialization, although the two are closely connected.
Why the Drug Discovery Process Is So Important
Effective drug discovery determines:
-
Whether a project has a strong scientific foundation
-
How safe and selective a compound is likely to be
-
Whether development costs can be justified
-
The probability of success in later clinical stages
A weak discovery phase often leads to costly failures during trials, while a well-executed process increases the likelihood of producing safe and effective therapies.
Core Stages of the Drug Discovery Process
Although specific workflows differ by organization and therapeutic area, most projects follow a common structure.
1. Disease Understanding and Target Identification
Drug discovery begins with understanding the biological mechanisms underlying a disease.
Researchers aim to identify:
-
Proteins or enzymes involved in disease progression
-
Genetic mutations linked to pathology
-
Signaling pathways that can be modulated therapeutically
The chosen target must be both biologically relevant and technically druggable.
2. Target Validation
Once identified, the target must be validated to confirm that modifying its activity produces a meaningful therapeutic effect.
Validation methods include:
-
Genetic knockdown or overexpression
-
Use of existing tool compounds
-
Animal disease models
-
Biomarker analysis
This step reduces the risk of pursuing targets that are scientifically interesting but clinically irrelevant.
3. Hit Discovery
In this stage, researchers search for compounds that interact with the validated target.
Common approaches include:
-
High-throughput screening of chemical libraries
-
Structure-based virtual screening
-
Fragment-based discovery
-
Natural product screening
Compounds that show measurable activity are called hits.
4. Hit to Lead Optimization
Hits are rarely suitable as drugs without further refinement.
Scientists modify their chemical structures to improve:
-
Potency
-
Selectivity
-
Solubility
-
Stability
-
Safety profile
This iterative process produces lead compounds with more favorable properties.
5. Lead Optimization
Lead compounds undergo extensive testing and chemical refinement.
Key goals include:
-
Improving absorption and distribution in the body
-
Reducing toxicity
-
Enhancing metabolic stability
-
Optimizing dosing characteristics
Multiple candidate molecules may be evaluated in parallel.
6. Preclinical Testing
Before entering clinical trials, promising candidates are tested in laboratory and animal models.
Preclinical studies evaluate:
-
Pharmacokinetics
-
Toxicology
-
Mechanism of action
-
Preliminary efficacy
Successful completion of this stage allows the compound to move into human testing.
Supporting Technologies in Modern Drug Discovery
The drug discovery process has evolved significantly due to technological advances.
Key tools include:
-
High-throughput screening platforms
-
Computational modeling and molecular docking
-
Genomics and proteomics
-
Artificial intelligence-assisted compound design
-
Automated synthesis and testing systems
These technologies increase efficiency and reduce the number of compounds required to find viable candidates.
Timelines and Costs
Drug discovery is a long-term investment.
Typical characteristics:
-
3 to 6 years for discovery and preclinical stages
-
Thousands of compounds screened
-
Only a small fraction advance to clinical trials
-
Costs reaching hundreds of millions of dollars
Because of these realities, companies carefully manage risk and prioritize projects with strong scientific justification.
Common Challenges in the Drug Discovery Process
Despite careful planning, many projects fail.
Major challenges include:
-
Poor target selection
-
Unexpected toxicity
-
Low bioavailability
-
Insufficient efficacy
-
Competitive market changes
Even with advanced tools, biological systems remain complex and unpredictable.
Strategies for Improving Success Rates
Organizations use several strategies to strengthen their drug discovery efforts.
-
Early integration of safety testing
-
Use of predictive biomarkers
-
Collaboration with academic research centers
-
Adoption of data-driven decision making
-
Portfolio diversification
These approaches help balance innovation with risk management.
Role of Outsourcing and Partnerships
Many companies collaborate with contract research organizations and academic institutions.
Benefits include:
-
Access to specialized expertise
-
Reduced infrastructure costs
-
Faster project timelines
-
Flexible resource allocation
Strategic partnerships are now a standard component of modern drug discovery.
Regulatory Considerations During Discovery
Although formal regulatory review occurs later, discovery teams must consider:
-
Data integrity requirements
-
Ethical use of animal models
-
Documentation standards
-
Intellectual property protection
Early compliance simplifies later development stages.
Applications Beyond Traditional Pharmaceuticals
The principles of the drug discovery process also apply to:
-
Biologic therapeutics
-
Gene therapies
-
Cell-based treatments
-
Vaccines
-
Diagnostic agents
Each category introduces unique technical and regulatory challenges but follows the same core scientific logic.
Impact on Patients and Healthcare Systems
Successful drug discovery leads to:
-
New treatment options
-
Improved survival rates
-
Better quality of life
-
Reduced long-term healthcare costs
Failures, while costly, also contribute valuable scientific knowledge that informs future research.
Best Practices for Managing Drug Discovery Projects
Experienced organizations follow consistent operational principles:
-
Define clear project milestones
-
Use standardized screening assays
-
Maintain transparent data management
-
Conduct regular portfolio reviews
-
Integrate cross-functional teams
These practices improve coordination and scientific rigor.
Conclusion
The Drug Discovery Process is the foundation of modern therapeutic innovation. It transforms fundamental biological insights into drug candidates with the potential to improve or save lives.
Although time-consuming and expensive, this structured approach allows researchers to systematically evaluate disease targets, optimize chemical compounds, and reduce the risks associated with clinical development. As technologies continue to evolve, the efficiency and precision of drug discovery will improve, accelerating the arrival of new treatments for patients worldwide.
For biotechnology companies, pharmaceutical developers, and research institutions, mastering this process is essential for long-term success in an increasingly competitive healthcare landscape.
Frequently Asked Questions
What are the main stages of the drug discovery process?
The main stages include target identification, target validation, hit discovery, lead optimization, and preclinical testing before a compound enters clinical trials.
How long does the drug discovery process usually take?
The discovery phase typically takes several years, often between three and six years, depending on the complexity of the disease and the technology used.
Why do many drug discovery projects fail?
Failures often occur due to lack of efficacy, unexpected toxicity, poor pharmacokinetics, or incorrect assumptions about disease biology.
How does drug discovery differ from drug development?
Drug discovery focuses on finding and optimizing candidate compounds, while drug development involves clinical trials, regulatory approval, manufacturing, and market launch.
Download PDF
Enter your email address to unlock the full PDF download.
Generating PDF...