Stem cell differentiation from bone marrow is a cornerstone of modern regenerative medicine and hematology. Bone marrow serves as one of the body’s most important reservoirs of adult stem cells, supplying specialized cells that maintain blood production, immune defense, and tissue repair throughout life. Understanding how these stem cells differentiate provides critical insight into disease treatment, transplantation therapy, and emerging biomedical technologies.
This article offers a comprehensive overview of how stem cell differentiation from bone marrow occurs, the types of stem cells involved, the factors that regulate the process, and the practical applications that are shaping current and future medical care.
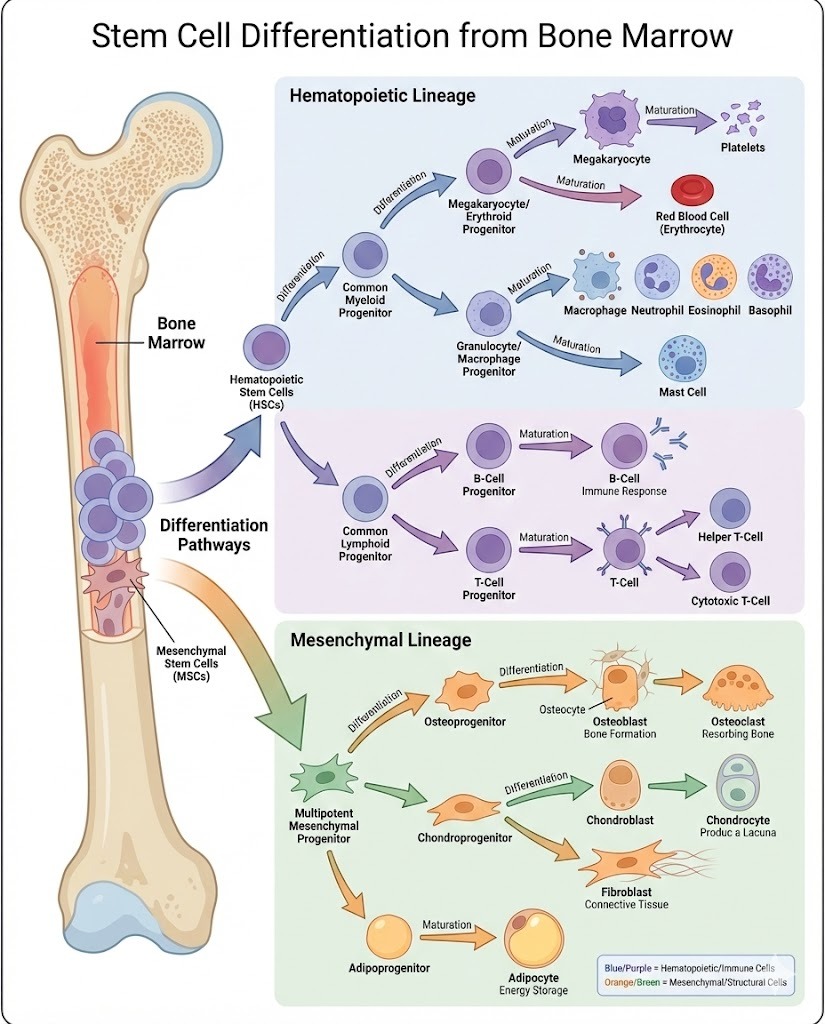
Understanding Bone Marrow Stem Cells
Bone marrow contains two primary categories of stem cells, each responsible for producing different types of specialized cells.
Hematopoietic stem cells
These stem cells generate all blood and immune system cells, including:
-
Red blood cells
-
White blood cells
-
Platelets
-
Specialized immune cells such as T cells and B cells
Hematopoietic stem cells are essential for oxygen transport, infection defense, and blood clotting.
Mesenchymal stem cells
Mesenchymal stem cells differentiate into non-blood tissues such as:
-
Bone cells
-
Cartilage cells
-
Fat cells
-
Muscle cells
-
Stromal cells that support organ structure
Together, these two stem cell populations drive the biological significance of stem cell differentiation from bone marrow.
What Is Stem Cell Differentiation?
Stem cell differentiation is the process by which unspecialized cells transform into mature cells with specific structures and functions. This transformation involves changes in gene expression, protein production, and cellular morphology.
In the context of bone marrow, differentiation ensures continuous replacement of aging or damaged cells and allows the body to respond to injury or infection.
This process is tightly regulated to maintain balance. Excessive or insufficient differentiation can result in serious disorders such as anemia, immune deficiency, or leukemia.
Biological Mechanisms Behind Differentiation
Stem cell differentiation from bone marrow is guided by a combination of internal genetic programs and external environmental signals.
Genetic regulation
Specific genes are activated or suppressed to determine a cell’s fate. Transcription factors play a central role by binding to DNA and initiating the expression of lineage-specific proteins.
Microenvironmental influence
The bone marrow niche provides physical and chemical support that influences stem cell behavior.
Important components include:
-
Growth factors and cytokines
-
Oxygen concentration
-
Cell-to-cell contact
-
Extracellular matrix proteins
Signal transduction pathways
Molecular signaling networks such as Wnt, Notch, and Hedgehog regulate whether a stem cell remains undifferentiated or begins transforming into a specialized cell type.
Epigenetic modifications
Chemical changes to DNA and histone proteins alter how genes are expressed without changing the underlying genetic code, allowing flexibility in differentiation outcomes.
Stages of Differentiation in Bone Marrow
Stem cell differentiation from bone marrow occurs in gradual stages rather than a single step.
-
Self-renewal phase
Stem cells divide to maintain the stem cell population. -
Commitment phase
Cells become restricted to a specific lineage such as blood or connective tissue. -
Progenitor stage
Cells rapidly divide and begin expressing markers of their future function. -
Maturation stage
Cells develop full functional capabilities and enter circulation or tissue environments.
This structured progression ensures stability and adaptability in cell production.
Clinical Applications
The medical importance of stem cell differentiation from bone marrow cannot be overstated. It forms the basis of many established and emerging therapies.
Bone marrow transplantation
One of the most well-known applications is hematopoietic stem cell transplantation for treating:
-
Leukemia
-
Lymphoma
-
Aplastic anemia
-
Genetic blood disorders
Transplanted stem cells repopulate the patient’s bone marrow and restore normal blood cell production.
Regenerative medicine
Mesenchymal stem cells are being studied for their ability to repair damaged tissues such as:
-
Bone fractures
-
Cartilage injuries
-
Spinal cord damage
-
Heart muscle after heart attacks
Immunotherapy
Bone marrow derived stem cells contribute to the development of engineered immune cells used in cancer treatments, including CAR T cell therapy.
Drug testing and disease modeling
Laboratory differentiation of bone marrow stem cells allows researchers to study disease mechanisms and evaluate new medications in controlled environments.
Advantages of Bone Marrow Derived Stem Cells
Several characteristics make bone marrow an attractive source for stem cell therapies.
-
Proven safety in clinical use
-
Ability to generate diverse cell types
-
Established harvesting techniques
-
Reduced ethical concerns compared to embryonic stem cells
-
Compatibility with autologous transplantation
These advantages support continued investment in bone marrow based regenerative technologies.
Challenges and Limitations
Despite its promise, stem cell differentiation from bone marrow faces technical and biological challenges.
-
Limited stem cell quantity in older individuals
-
Risk of immune rejection in donor transplants
-
Difficulty controlling differentiation pathways precisely
-
Potential for abnormal growth or tumor formation
-
High cost of clinical procedures
Ongoing research aims to improve efficiency, safety, and accessibility.
Emerging Research Directions
Scientific advances continue to refine understanding and practical use of bone marrow stem cells.
Key areas of innovation include:
-
Gene editing to correct inherited disorders
-
3D tissue scaffolds for improved cell integration
-
Personalized stem cell therapies
-
Artificial bone marrow environments
-
Automated cell manufacturing systems
These developments may dramatically expand therapeutic options over the next decade.
Ethical and Regulatory Considerations
Because bone marrow stem cells are obtained from adult donors, ethical concerns are relatively limited compared to embryonic sources. However, strict regulatory oversight governs:
-
Donor consent
-
Cell processing standards
-
Clinical trial design
-
Patient safety monitoring
Compliance ensures responsible medical application.
Conclusion
Stem cell differentiation from bone marrow represents one of the most powerful biological processes supporting human health and medical innovation. By continuously generating blood cells and contributing to tissue repair, bone marrow stem cells sustain vital physiological functions throughout life.
Advances in understanding the mechanisms behind differentiation are unlocking new therapies for cancer, immune disorders, and degenerative diseases. While challenges remain, the combination of proven clinical success and rapid technological progress positions bone marrow derived stem cells as a central pillar of modern regenerative medicine.
As research evolves, stem cell differentiation from bone marrow will continue to shape the future of personalized treatment and tissue engineering.
Frequently Asked Questions About Stem Cell Differentiation from Bone Marrow
What is stem cell differentiation from bone marrow?
Stem cell differentiation from bone marrow is the process by which bone marrow stem cells transform into specialized blood, immune, or connective tissue cells needed for normal body function and repair.
Which types of stem cells are found in bone marrow?
Bone marrow contains hematopoietic stem cells that form blood and immune cells and mesenchymal stem cells that produce bone, cartilage, fat, and supportive tissues.
How is bone marrow stem cell differentiation used in medicine?
It is widely used in bone marrow transplantation for cancer treatment, regenerative therapies for tissue repair, and the development of advanced immunotherapies.
Is stem cell differentiation from bone marrow safe?
When performed under regulated clinical conditions, it has a strong safety record. Risks include infection, immune reactions, and graft failure, but medical protocols are designed to minimize these complications.