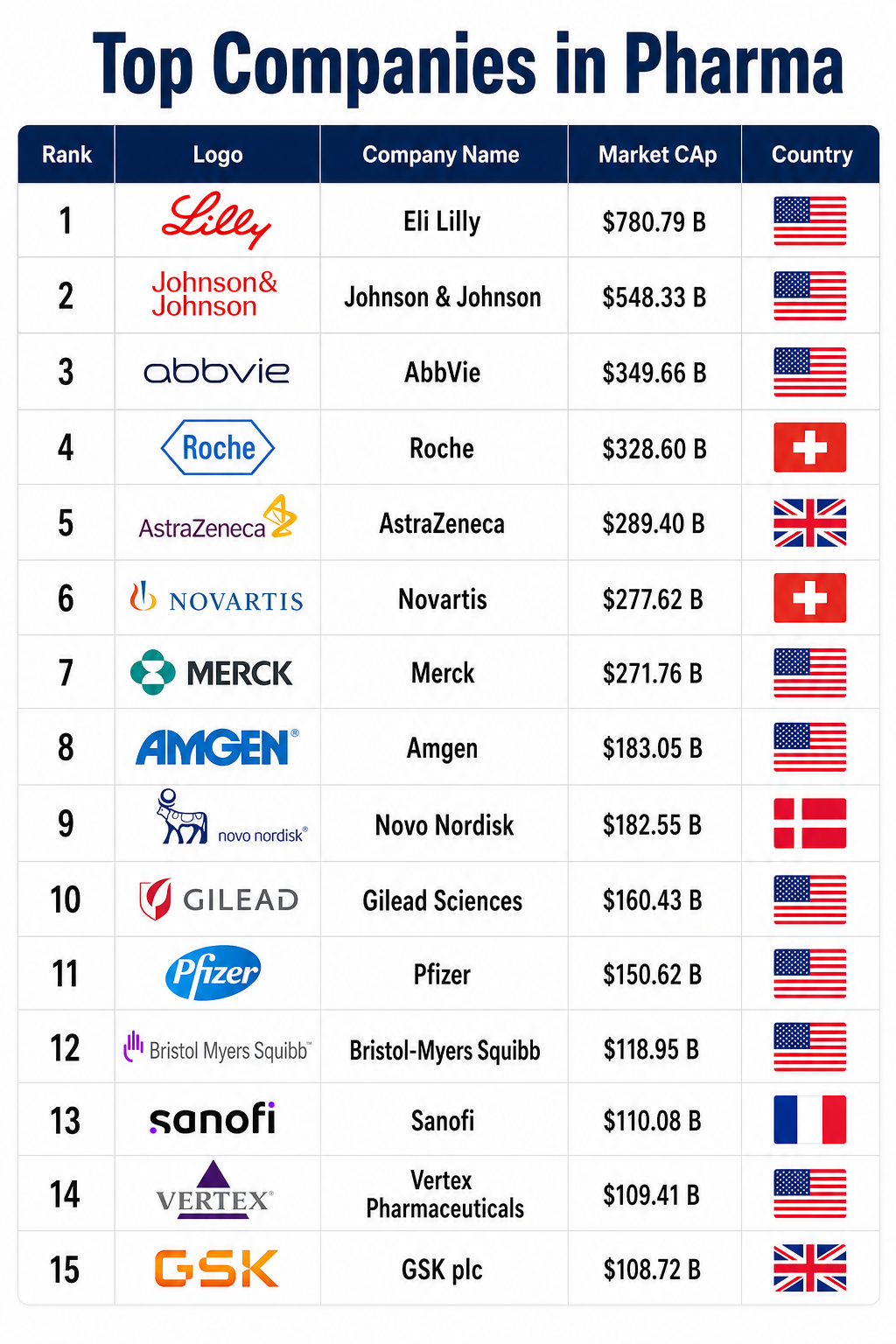
The pharmaceutical industry in 2026 stands at a crossroads of radical technological integration and unprecedented regulatory volatility. After a period of intense front-loading and pandemic-induced urgency, the sector has matured into a more calculated, data-driven, and “resilient” ecosystem. The global pharmaceutical market is projected to reach approximately $2.15 trillion this year, with North America maintaining a dominant 44% share, even as the Asia-Pacific region accelerates as the world’s fastest-growing hub. However, the narrative of 2026 is not just about market size; it is about the “Agentic Era” of drug discovery and the geopolitical restructuring of how medicines are produced and priced.
The Geopolitical Chessboard: Reshoring and “U.S.-First” Policy
The U.S. landscape has undergone a tectonic shift since 2025. A new administration has introduced aggressive protectionist measures, including roughly 15% tariffs on pharmaceutical imports and “U.S.-first” production incentives. While exemptions exist for certain generics and negotiated branded drugs, these policies have added an estimated $13 billion to $19 billion in industry costs. This “reshoring” push has incentivized the construction of domestic facilities, but the high cost of labor and materials remains a friction point. Simultaneously, a new pricing framework—often referred to in policy circles as a “most-favored-nation” (MFN) model—is exerting downward pressure on drug prices by indexing U.S. costs to international benchmarks. This has forced major pharmaceutical companies to forge direct agreements with the government to secure tariff relief in exchange for price caps, fundamentally altering the traditional revenue models for blockbusters.
The Rise of Agentic AI and Digital Twins
Technologically, 2026 marks the transition from “AI as a tool” to Agentic AI as a core workforce component. Generative AI models are no longer just summarizing data; they are simulating how new molecules interact within the human body, effectively compressing drug development timelines from years to months. Smart factories are now the industry standard, utilizing Digital Twin technology to replicate production environments in a virtual space. Companies like Eli Lilly and Johnson & Johnson are using these twins to predict equipment failure and run “what-if” scenarios for complex manufacturing processes without halting actual lines. Furthermore, “Ambient AI” has revolutionized clinical trials and provider interactions. AI scribes now handle the bulk of administrative documentation, allowing clinicians to focus on patient-centric care. The integration of genomic data with real-time feedback from personal wearable devices has also enabled a new level of precision medicine, where Alzheimer’s or kidney diseases can be predicted years before the first symptom appears.
Therapeutic Frontiers: Biologics and ADCs
The drug pipeline in 2026 is heavily skewed toward large-molecule biopharmaceuticals. Biologics and biosimilars now account for nearly 40% of the total drug pipeline and are projected to hold over 75% of the market share by molecule type this year. Oncology remains the dominant therapeutic area, representing roughly 17.6% of the market. Antibody-drug conjugates (ADCs) have seen a massive resurgence; after a brief hiatus in the early 2020s, several novel ADCs were approved in late 2025 and early 2026, offering more targeted, less toxic chemotherapy alternatives. There is also a significant consolidation of TIDES (peptides and oligonucleotides). The FDA’s recent approvals highlight a trend toward liver-targeted delivery systems using GalNAc technology, which allows for highly specific genetic silencing or enhancement.
Supply Chain Resilience and Regulatory Tightening
Supply chains in 2026 are governed by the full implementation of the Drug Supply Chain Security Act (DSCSA). This requires total serialization and interoperability, meaning every single unit of medicine is tracked from the factory floor to the patient’s hand. While this has drastically reduced the presence of counterfeit drugs, it has also created a massive “digital attack surface.” Cyber insurance has become as critical as product liability insurance, as a single ransomware attack on a serialized database can now paralyze an entire national supply chain. On the regulatory front, the FDA and EMA have tightened their requirements for cell and gene therapies, placing a heavy emphasis on CMC (Chemistry, Manufacturing, and Controls). Developers are now expected to have scalable, globally viable manufacturing plans even at the pre-IND stage, as the agencies move away from “accelerated” pathways that lack long-term lifecycle management.
The Patient as a Prosumer
Finally, 2026 has seen the “patient in the driver’s seat.” Fed up with traditional intermediaries, more pharmaceutical companies are building direct-to-patient (DTP) relationships. Digital portals and holistic wellness apps are being integrated into the prescription itself, helping patients manage sleep, nutrition, and exercise alongside their medication. This shift is turning patients into “prosumers”—proactive consumers of their own health data—who demand transparency in pricing and clinical efficacy. In summary, the 2026 pharmaceutical scenario is defined by a paradox: it is an industry that is becoming more localized through domestic manufacturing and tariffs, yet more globalized through the universal language of AI and biotechnology. Success no longer belongs to the company with the largest sales force, but to the one with the most integrated data architecture and the agility to navigate a volatile regulatory ocean.