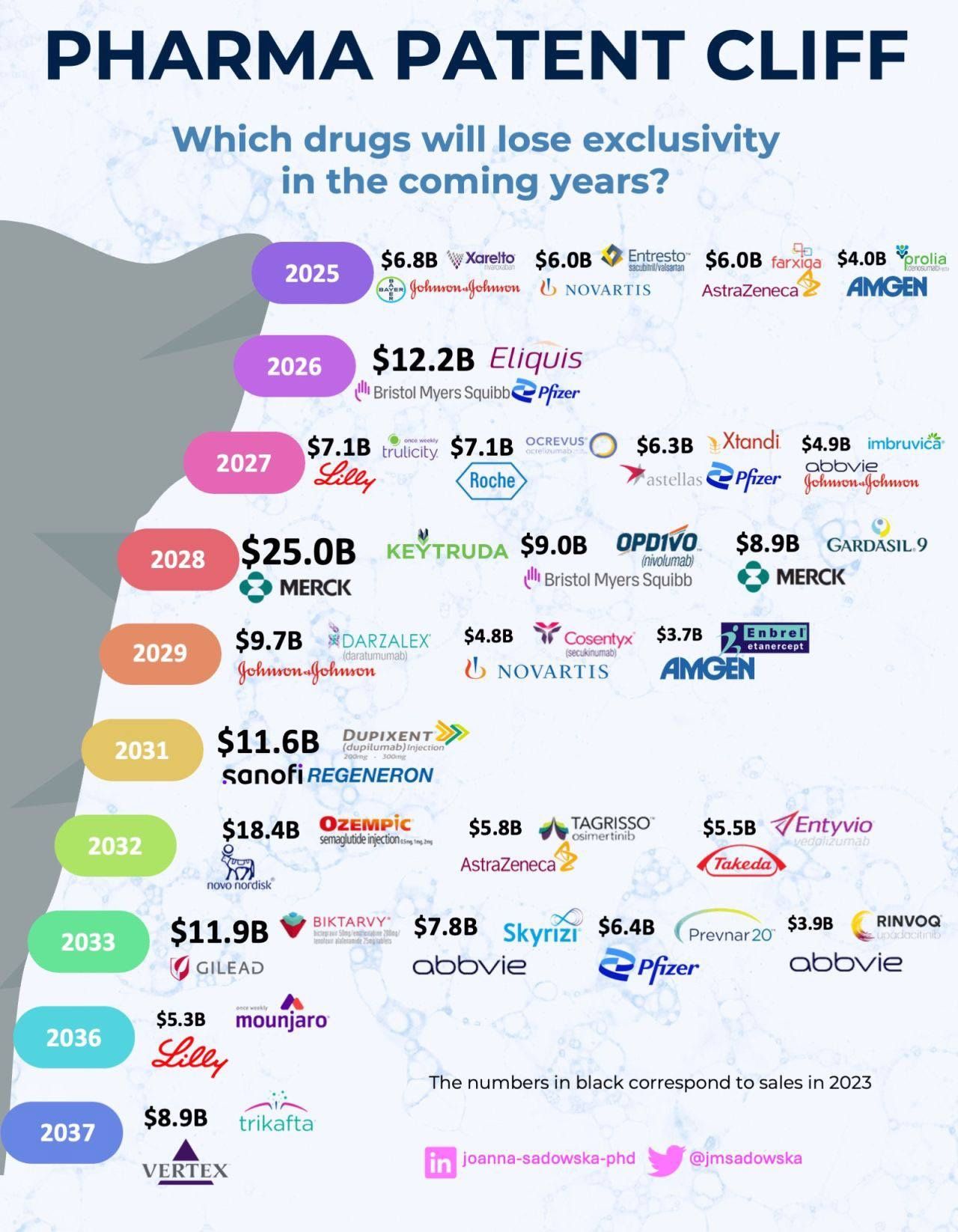
The global pharmaceutical industry is approaching a major patent cliff, with several blockbuster drugs expected to lose market exclusivity over the next decade. Patent expirations open the door to generic and biosimilar competition, often triggering sharp revenue declines for originator companies while reshaping pricing, access, and competitive dynamics.
Based on 2023 sales figures highlighted in the infographic, the upcoming patent cliff represents tens of billions of dollars in at-risk revenue, particularly across oncology, immunology, cardiology, and metabolic disease segments.
2025: Early Signals of Revenue Erosion
In 2025, several high-revenue therapies are expected to face exclusivity loss:
-
Xarelto ($6.8B) – Johnson & Johnson
-
Entresto ($6.0B) – Novartis
-
Farxiga ($6.0B) – AstraZeneca
-
Prolia ($4.0B) – Amgen
These therapies span cardiovascular, diabetes, and osteoporosis indications, making 2025 an important transition year for chronic disease markets.
2026: A Major Hit from Eliquis
-
Eliquis ($12.2B) – Bristol Myers Squibb and Pfizer
The loss of exclusivity for Eliquis represents one of the largest single patent expirations of the decade, with significant implications for the global anticoagulants market.
2027: Diabetes, Oncology, and Neurology Under Pressure
Key products facing patent expiry include:
-
Trulicity ($7.1B) – Eli Lilly
-
Ocrevus ($7.1B) – Roche
-
Xtandi ($6.3B) – Pfizer / Astellas
-
Imbruvica ($4.9B) – AbbVie / Johnson & Johnson
This year highlights increasing biosimilar risk in oncology and autoimmune therapies.
2028: The Largest Patent Cliff Event
-
Keytruda ($25.0B) – Merck
-
Opdivo ($9.0B) – Bristol Myers Squibb
-
Gardasil 9 ($8.9B) – Merck
The loss of exclusivity for Keytruda alone marks the single most significant patent cliff event, potentially redefining the global oncology treatment landscape.
2029–2031: Immunology and Dermatology Take Center Stage
-
Darzalex ($9.7B) – Johnson & Johnson
-
Cosentyx ($4.8B) – Novartis
-
Enbrel ($3.7B) – Amgen
-
Dupixent ($11.6B) – Sanofi / Regeneron
These expirations will intensify competition in autoimmune and inflammatory disease markets, where biosimilars are already gaining traction.
2032–2033: Metabolic and Infectious Disease Impact
-
Ozempic ($18.4B) – Novo Nordisk
-
Tagrisso ($5.8B) – AstraZeneca
-
Entyvio ($5.5B) – Takeda
-
Biktarvy ($11.9B) – Gilead
-
Skyrizi ($7.8B) – AbbVie
-
Prevnar 20 ($6.4B) – Pfizer
-
Rinvoq ($3.9B) – AbbVie
The expiration of Ozempic underscores looming challenges in the GLP-1 and obesity treatment market, one of the fastest-growing segments in pharma.
2036–2037: Long-Tail Blockbusters
-
Mounjaro ($5.3B) – Eli Lilly
-
Trikafta ($8.9B) – Vertex
These later expirations demonstrate how long-lifecycle specialty drugs continue to anchor revenues well into the 2030s.
What the Patent Cliff Means for the Industry
The upcoming pharma patent cliff will:
-
Accelerate generic and biosimilar adoption
-
Increase pricing pressure globally
-
Drive M&A, lifecycle management, and innovation
-
Shift focus toward next-generation biologics and precision therapies
For pharmaceutical companies, proactive portfolio diversification and R&D productivity will be critical. For payers and patients, the patent cliff promises greater access and affordability.