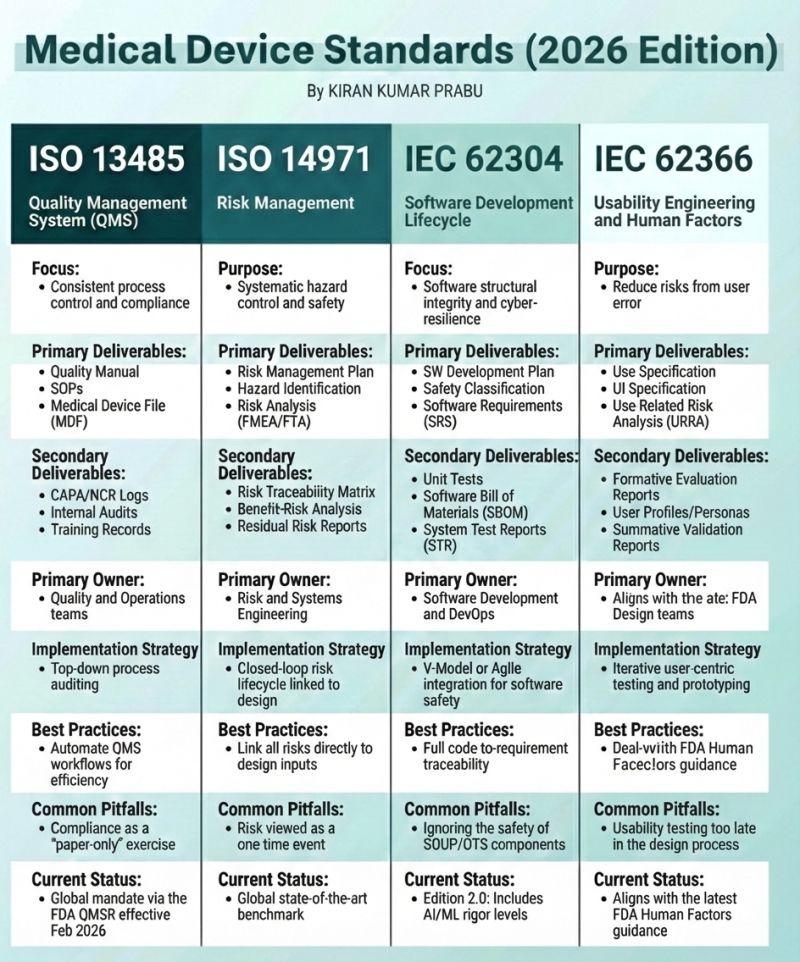
As medical devices become more software-driven and user-centric, regulatory expectations are rising fast. The 2026 edition of global medical device standards highlights four critical frameworks that every manufacturer, startup, and compliance professional must understand.
These standards don’t work in isolation—they form a connected ecosystem covering quality, risk, software safety, and usability.
Let’s break them down one by one.
ISO 13485 – Quality Management System (QMS)
Focus:
Ensuring consistent process control and regulatory compliance across the product lifecycle.
Why it matters:
ISO 13485 is the backbone of medical device compliance. It ensures that organizations can consistently design, manufacture, and deliver safe medical devices.
Primary Deliverables
-
Quality Manual
-
Standard Operating Procedures (SOPs)
-
Medical Device File (MDF)
Secondary Deliverables
-
CAPA & NCR logs
-
Internal audit reports
-
Training records
Primary Owner:
Quality and Operations teams
Implementation Strategy:
Top-down, process-driven auditing
Best Practice:
Automate QMS workflows to improve efficiency and traceability.
Common Pitfall:
Treating compliance as a “paper-only” exercise instead of a living system.
Current Status (2026):
Globally mandated, with FDA QMSR effective from February 2026.
ISO 14971 – Risk Management
Purpose:
Systematic identification, analysis, and control of risks throughout the device lifecycle.
Why it matters:
Risk management is not a one-time task—it must be continuously linked to design, development, and post-market surveillance.
Primary Deliverables
-
Risk Management Plan
-
Hazard Identification
-
Risk Analysis (FMEA / FTA)
Secondary Deliverables
-
Risk Traceability Matrix
-
Benefit-Risk Analysis
-
Residual Risk Reports
Primary Owner:
Risk and Systems Engineering teams
Implementation Strategy:
Closed-loop risk lifecycle tightly integrated with design.
Best Practice:
Directly link all risks to design inputs and controls.
Common Pitfall:
Viewing risk management as a single event instead of an ongoing process.
Current Status (2026):
Considered the global state-of-the-art benchmark.
IEC 62304 – Software Development Lifecycle
Focus:
Software structural integrity, safety, and cyber resilience.
Why it matters:
With software now central to most medical devices, IEC 62304 ensures disciplined, traceable, and safe software development.
Primary Deliverables
-
Software Development Plan
-
Safety Classification
-
Software Requirements Specification (SRS)
Secondary Deliverables
-
Unit test records
-
Software Bill of Materials (SBOM)
-
System Test Reports (STR)
Primary Owner:
Software Development and DevOps teams
Implementation Strategy:
V-Model or Agile methods adapted for software safety.
Best Practice:
Maintain full code-to-requirement traceability.
Common Pitfall:
Ignoring safety risks from SOUP/OTS (third-party) software components.
Current Status (2026):
Edition 2.0, including AI/ML rigor levels.
IEC 62366 – Usability Engineering & Human Factors
Purpose:
Reducing risks caused by user error through better design.
Why it matters:
Many device failures are not technical—but usability related. This standard ensures devices are safe and intuitive for real users.
Primary Deliverables
-
Use Specification
-
UI Specification
-
Use-Related Risk Analysis (URRA)
Secondary Deliverables
-
Formative evaluation reports
-
User profiles and personas
-
Summative validation reports
Primary Owner:
Design teams (aligned with FDA expectations)
Implementation Strategy:
Iterative, user-centric testing and prototyping.
Best Practice:
Align closely with FDA Human Factors guidance.
Common Pitfall:
Conducting usability testing too late in the design process.
Current Status (2026):
Fully aligned with the latest FDA Human Factors guidance.
How These Standards Work Together
In 2026, regulators expect integration—not silos:
-
ISO 13485 defines how your organization works
-
ISO 14971 defines how risks are identified and controlled
-
IEC 62304 ensures software safety and reliability
-
IEC 62366 ensures safe and intuitive user interaction
Together, they create a complete compliance framework—from design to deployment and beyond.
Final Thought
Medical device compliance in 2026 is no longer about documentation alone. It’s about traceability, usability, risk awareness, and software discipline—all working together.
Organizations that embed these standards into their culture, not just their files, will move faster, safer, and with far fewer regulatory surprises.