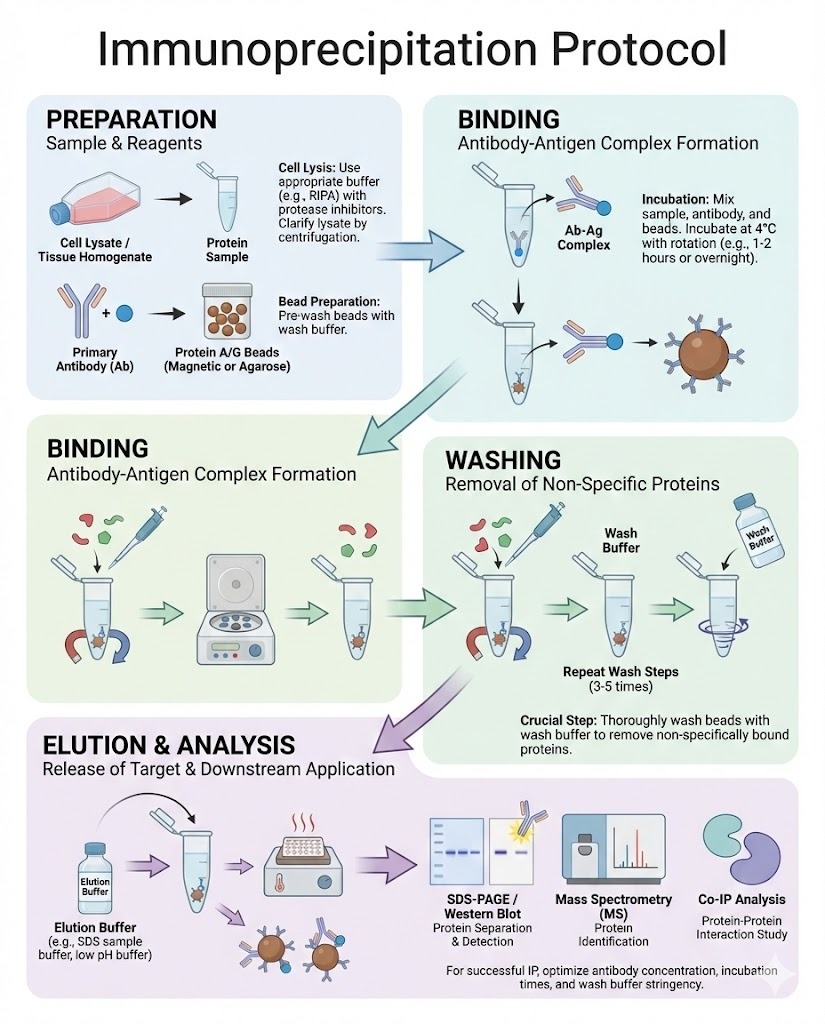
An immunoprecipitation protocol is a foundational laboratory method used to isolate and study specific proteins from complex biological samples. It plays a critical role in molecular biology, biochemistry, and biomedical research, enabling scientists to investigate protein expression, protein–protein interactions, and post translational modifications with high specificity.
Because proteins rarely function in isolation, immunoprecipitation provides a powerful way to examine biological pathways and disease mechanisms at the molecular level. This guide explains how the technique works, outlines the essential steps of a reliable immunoprecipitation protocol, and highlights best practices for generating reproducible, high quality results.
What Is Immunoprecipitation
Immunoprecipitation is an antibody based technique that selectively captures a target protein from a mixture such as cell lysate or tissue extract. The antibody binds to the protein of interest, forming a complex that can be separated from the rest of the sample using solid supports such as agarose or magnetic beads.
This process allows researchers to:
-
Enrich low abundance proteins
-
Analyze protein interactions
-
Detect post translational modifications
-
Validate antibody specificity
-
Prepare samples for downstream assays
Common downstream applications include Western blotting, mass spectrometry, enzyme activity assays, and structural analysis.
Types of Immunoprecipitation
Different experimental goals require different variations of the immunoprecipitation protocol.
Standard Immunoprecipitation
Used to isolate a single protein from a sample.
Co Immunoprecipitation
Designed to study protein–protein interactions by pulling down complexes.
Chromatin Immunoprecipitation
Targets proteins bound to DNA to study gene regulation.
RNA Immunoprecipitation
Used to isolate RNA binding proteins and their associated RNA molecules.
Although the basic workflow remains similar, each variation requires protocol adjustments.
Core Components of an Immunoprecipitation Protocol
A successful immunoprecipitation experiment depends on several critical elements.
-
High quality specific antibody
-
Appropriate lysis buffer
-
Protein binding beads
-
Proper washing conditions
-
Elution strategy
-
Suitable detection method
Optimizing each component improves specificity and yield.
Step by Step Immunoprecipitation Protocol Overview
While specific reagents and volumes vary by experiment, most protocols follow this general structure.
Sample Preparation and Cell Lysis
Cells or tissues are lysed to release proteins while preserving their structure and interactions.
Important considerations:
-
Use non denaturing buffers for interaction studies
-
Include protease and phosphatase inhibitors
-
Maintain cold temperatures
-
Avoid excessive mechanical disruption
The goal is to extract proteins without degrading them.
Pre Clearing the Lysate
Pre clearing removes proteins that bind nonspecifically to beads.
This step reduces background noise and improves specificity.
Antibody Incubation
The lysate is incubated with a specific antibody against the target protein.
Key variables include:
-
Antibody concentration
-
Incubation time
-
Temperature
-
Sample volume
Longer incubation at lower temperatures often improves binding efficiency.
Capture with Beads
Protein A, Protein G, or antibody coated beads bind the antibody–protein complex.
Magnetic beads are increasingly popular due to ease of handling and reduced sample loss.
Washing Steps
Multiple washes remove unbound proteins and contaminants.
Wash buffer composition balances:
-
Stringency to reduce background
-
Gentleness to preserve protein complexes
Typically three to five washes are performed.
Elution of Target Protein
The protein complex is released from the beads using:
-
Low pH buffers
-
SDS sample buffer
-
Competitive peptides
The eluted sample is ready for downstream analysis.
Key Factors That Affect Protocol Performance
Antibody Quality
Specificity and affinity directly determine success. Poor antibodies increase background and false positives.
Lysis Buffer Composition
Detergents and salt concentration influence protein solubility and interactions.
Bead Selection
Protein A and Protein G differ in species and antibody subclass affinity.
Incubation Conditions
Time and temperature affect binding kinetics.
Wash Stringency
Overwashing reduces yield. Underwashing increases contamination.
Balancing these factors is essential for reproducible results.
Common Applications in Research
An optimized immunoprecipitation protocol supports a wide range of studies.
-
Identification of signaling pathway components
-
Validation of protein interactions
-
Detection of phosphorylation or ubiquitination
-
Study of transcription factor complexes
-
Drug target validation
-
Biomarker discovery
In clinical research, immunoprecipitation contributes to understanding cancer biology, autoimmune disease mechanisms, and neurodegenerative disorders.
Troubleshooting Common Problems
Low Protein Yield
Possible causes:
-
Insufficient antibody
-
Low expression of target protein
-
Incomplete lysis
-
Overly harsh washing
High Background Signal
Common reasons:
-
Nonspecific antibody binding
-
Inadequate pre clearing
-
Insufficient washing
-
Cross reactive secondary antibodies
Loss of Protein Interactions
Caused by:
-
Strong detergents
-
High salt concentrations
-
Prolonged incubation at room temperature
Systematic optimization usually resolves these issues.
Best Practices for Reliable Results
To maximize consistency and data quality:
-
Use validated antibodies whenever possible
-
Include negative controls such as IgG controls
-
Keep samples cold
-
Standardize buffer compositions
-
Record incubation times precisely
-
Run technical replicates
-
Confirm results using independent methods
Documentation of protocol details improves reproducibility across laboratories.
Commercial Kits vs Custom Protocols
Many laboratories use commercial immunoprecipitation kits for convenience.
Advantages of kits:
-
Pre optimized reagents
-
Reduced setup time
-
Standardized performance
Advantages of custom protocols:
-
Greater flexibility
-
Cost efficiency for large studies
-
Ability to tailor conditions for specific proteins
The choice depends on experimental scale, budget, and technical expertise.
Importance in Modern Biomedical Research
The immunoprecipitation protocol remains indispensable despite advances in proteomics and high throughput screening. It offers unmatched specificity and versatility for probing molecular interactions in complex biological systems.
Its compatibility with mass spectrometry and genomic technologies further expands its relevance in systems biology and translational medicine.
Conclusion
A well designed immunoprecipitation protocol is a powerful tool for isolating proteins and studying molecular interactions within biological systems. By combining high quality antibodies, optimized buffers, controlled incubation conditions, and rigorous washing steps, researchers can achieve precise and reproducible protein enrichment.
Whether used for basic research, drug discovery, or disease mechanism studies, immunoprecipitation remains a cornerstone technique in molecular biology laboratories. Mastery of this protocol enhances experimental reliability and deepens understanding of cellular function at the protein level.
Frequently Asked Questions
What is an immunoprecipitation protocol used for
It is used to isolate specific proteins from complex samples to study protein expression, interactions, and modifications.
How long does a typical immunoprecipitation protocol take
Most experiments require four to eight hours, including sample preparation, antibody incubation, washing, and elution steps.
What is the difference between immunoprecipitation and co immunoprecipitation
Standard immunoprecipitation isolates a single target protein, while co immunoprecipitation captures protein complexes to study interactions.
Why is antibody selection critical in an immunoprecipitation protocol
The antibody determines binding specificity and efficiency. Poor antibody quality leads to low yield, high background, and unreliable results.
Download PDF
Enter your email address to unlock the full PDF download.
Generating PDF...