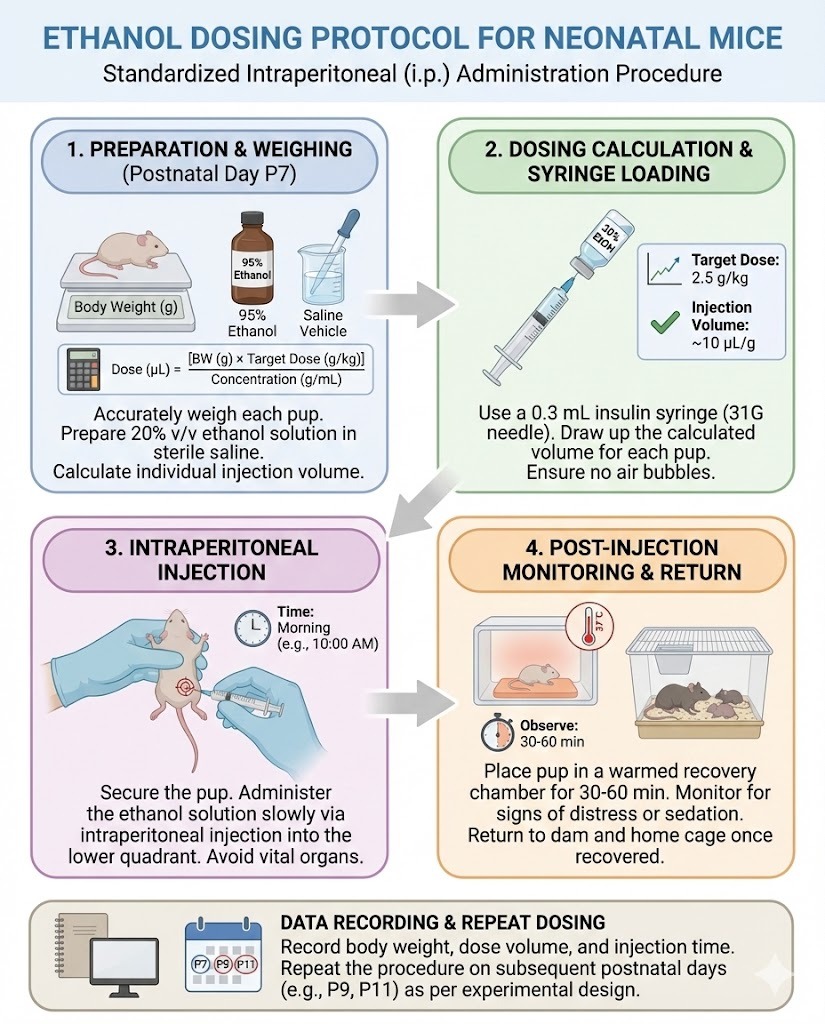
Research involving early-life exposure models requires exceptional care in experimental design, documentation, and ethical oversight. One commonly discussed framework in developmental neuroscience and toxicology is the Ethanol Dosing Protocol for Neonatal Mice, which is used to study how controlled alcohol exposure affects growth, brain development, and long-term behavior.
Rather than being a single universal recipe, this protocol is a structured approach that defines how ethanol is administered, monitored, and evaluated in neonatal animals under tightly regulated laboratory conditions. Understanding its components helps researchers design reproducible studies, maintain animal welfare standards, and generate reliable data that can be compared across institutions.
This article explains what the protocol involves, how it is typically structured, and what best practices are followed in professional research environments.
What Is an Ethanol Dosing Protocol for Neonatal Mice?
An ethanol dosing protocol for neonatal mice is a documented experimental framework that specifies:
-
The developmental window of exposure
-
The method of ethanol administration
-
Monitoring and welfare criteria
-
Data collection and analysis procedures
-
Ethical and regulatory safeguards
These protocols are commonly used in studies related to:
-
Fetal alcohol spectrum disorders
-
Neurodevelopmental toxicology
-
Metabolic programming
-
Behavioral neuroscience
Because neonatal mice are highly sensitive to environmental stressors, the protocol design must balance scientific objectives with strict animal welfare requirements.
Why Protocol Design Matters
Small variations in dosing strategy, timing, or handling can significantly alter experimental outcomes. A standardized ethanol dosing protocol for neonatal mice helps ensure:
-
Reproducibility between experiments
-
Reduced variability within study groups
-
Accurate interpretation of dose–response relationships
-
Compliance with institutional and regulatory guidelines
Poorly designed protocols increase the risk of confounding factors such as dehydration, nutritional stress, or maternal separation effects.
Core Elements of an Ethanol Dosing Protocol for Neonatal Mice
Although exact implementation details differ between laboratories, most protocols share several conceptual stages.
1. Study Planning and Ethical Approval
Before any experimental work begins, researchers define:
-
Scientific objectives and hypotheses
-
Target developmental stages
-
Outcome measures such as brain morphology or behavior
-
Sample size and statistical power
Ethical approval from an institutional animal care and use committee is mandatory. Protocol documentation must clearly justify the model and describe welfare safeguards.
2. Selection of Animal Model
Common considerations include:
-
Mouse strain and genetic background
-
Litter size standardization
-
Sex distribution
-
Age in postnatal days
Neonatal mice represent a developmental stage roughly equivalent to late gestation in humans, making them relevant for specific neurodevelopmental questions.
3. Administration Strategy
The administration strategy defines how ethanol is delivered. While details vary by institution, the protocol typically specifies:
-
Route of administration
-
Frequency and duration of exposure
-
Timing relative to circadian cycle
-
Handling procedures to minimize stress
The goal is to achieve controlled systemic exposure while minimizing confounding physiological stress.
4. Control Groups
A robust ethanol dosing protocol for neonatal mice always includes appropriate controls, such as:
-
Untreated animals
-
Vehicle-treated animals
-
Pair-fed or handling-matched controls
These groups help distinguish ethanol-specific effects from general procedural stress.
5. Monitoring and Welfare Assessment
Continuous monitoring is central to ethical compliance and data quality. Protocols usually define:
-
Body weight tracking
-
Hydration and feeding behavior
-
Skin color and activity levels
-
Developmental milestones
Humane endpoints are predetermined to prevent unnecessary suffering and to maintain scientific integrity.
6. Sample Collection and Data Recording
Depending on the study objectives, data collection may include:
-
Blood or tissue sampling
-
Brain imaging or histology
-
Behavioral testing
-
Molecular assays
Standardized documentation practices ensure that results can be audited and replicated.
Variables That Influence Experimental Outcomes
Even when following a formal ethanol dosing protocol for neonatal mice, multiple variables can affect results.
Key factors include:
-
Genetic strain differences
-
Litter effects
-
Environmental temperature and humidity
-
Maternal care patterns
-
Handling frequency
Experienced laboratories account for these factors in their study design and statistical analysis.
Regulatory and Ethical Considerations
Research involving neonatal animals is subject to heightened scrutiny. Protocols must align with:
-
National animal welfare legislation
-
Institutional ethical review boards
-
International guidelines such as the 3Rs principle
The 3Rs emphasize:
-
Replacement with alternative models when possible
-
Reduction in the number of animals used
-
Refinement of techniques to minimize distress
These principles are integral to any professionally developed ethanol dosing protocol for neonatal mice.
Common Applications in Research
Well-designed protocols support a wide range of scientific investigations.
Typical applications include:
-
Studying structural brain development
-
Identifying molecular pathways affected by early ethanol exposure
-
Evaluating potential therapeutic interventions
-
Assessing long-term cognitive and behavioral outcomes
Because early developmental stages are highly sensitive, results from these studies often provide insights that are difficult to obtain from adult models.
Documentation and Reproducibility
Modern research standards emphasize transparency. A complete protocol document usually includes:
-
Rationale for experimental design
-
Detailed timelines
-
Welfare monitoring criteria
-
Statistical analysis plans
-
Data storage methods
This level of documentation allows other researchers to interpret findings correctly and assess methodological rigor.
Best Practices for Protocol Implementation
Laboratories with consistent results tend to follow several shared practices:
-
Comprehensive training for animal handlers
-
Routine equipment calibration
-
Pilot studies to refine procedures
-
Regular protocol review and updates
-
Independent welfare audits
These measures improve both ethical compliance and data reliability.
Conclusion
The Ethanol Dosing Protocol for Neonatal Mice is not simply a technical procedure but a structured research framework that integrates experimental design, animal welfare, and data integrity. When carefully planned and responsibly implemented, it enables researchers to explore how early-life ethanol exposure influences development while maintaining high ethical standards.
For institutions conducting developmental or toxicological research, investing time in protocol design, staff training, and documentation is essential. These efforts improve reproducibility, protect animal well-being, and strengthen the scientific value of the results.
Frequently Asked Questions
What is the purpose of an ethanol dosing protocol for neonatal mice?
The protocol is designed to standardize how ethanol exposure is studied during early development, allowing researchers to investigate biological and behavioral effects while maintaining ethical and methodological consistency.
How is an ethanol exposure study in neonatal mice typically structured?
Most studies define a specific developmental window, an administration strategy, monitoring procedures, and clear outcome measures, all documented within an approved protocol.
Why are control groups important in neonatal ethanol dosing studies?
Control groups help distinguish the effects of ethanol from stress related to handling, nutrition, or environmental conditions, improving the reliability of conclusions.
Can ethanol dosing protocols for neonatal mice differ between laboratories?
Yes. Protocols vary based on research goals, institutional guidelines, and regulatory requirements, but they generally follow the same conceptual framework of planning, controlled exposure, monitoring, and analysis.