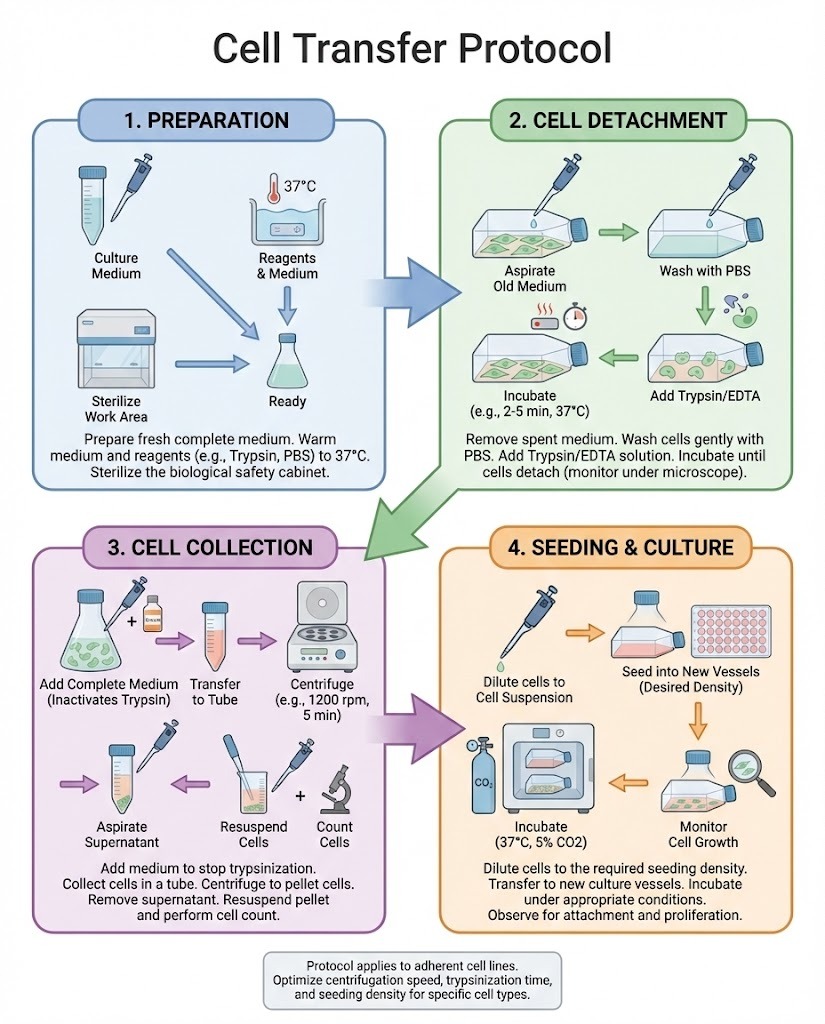
Cell-based experiments depend heavily on how cells are handled, moved, and maintained between vessels, instruments, or experimental stages. A well-defined Cell Transfer Protocol ensures that cells remain viable, uncontaminated, and physiologically stable throughout laboratory procedures. Whether working in basic research, biotechnology development, or clinical manufacturing, standardized transfer methods are essential for reliable results.
This guide explains what a cell transfer protocol is, why it matters, how it is typically structured, and which best practices help laboratories achieve consistent outcomes.
What Is a Cell Transfer Protocol?
A cell transfer protocol is a documented set of procedures describing how cells are moved from one environment to another while preserving their integrity and function. Transfers may occur between:
-
Culture flasks and multiwell plates
-
Bioreactors and storage containers
-
Centrifuge tubes and assay systems
-
Donor and recipient systems in experimental models
The protocol outlines each step, including preparation, handling techniques, environmental controls, and post-transfer monitoring.
Why Cell Transfer Protocols Are Critical
Cells are sensitive to physical stress, temperature fluctuations, osmotic changes, and contamination. Improper handling during transfer can lead to:
-
Reduced cell viability
-
Altered gene expression
-
Changes in morphology or differentiation status
-
Experimental variability
-
Microbial contamination
A standardized cell transfer protocol minimizes these risks and ensures that experimental results reflect biological phenomena rather than technical artifacts.
Core Objectives of a Cell Transfer Protocol
Most protocols are designed to achieve several fundamental goals:
-
Maintain cell viability and functionality
-
Prevent microbial contamination
-
Preserve sterility and traceability
-
Reduce mechanical and thermal stress
-
Ensure reproducibility across experiments and operators
Meeting these objectives is especially important in regulated environments such as clinical research and cell therapy manufacturing.
Common Applications of Cell Transfer Protocols
Cell transfer procedures are used across many scientific and industrial fields.
Typical applications include:
-
Routine passaging of cell cultures
-
Preparation of cells for transfection or drug treatment
-
Transfer between growth phases in bioprocessing
-
Cryopreservation and recovery workflows
-
In vivo transplantation studies
-
Quality control testing
Each application may require minor adjustments, but the underlying principles remain consistent.
Key Components of a Standard Cell Transfer Protocol
While specific details vary depending on cell type and laboratory setting, most protocols include the following stages.
1. Preparation of Materials and Workspace
Before handling cells, laboratories prepare:
-
Sterile culture vessels
-
Appropriate growth media or buffers
-
Pipettes and tips
-
Centrifuge tubes
-
Personal protective equipment
Work is typically performed in a biosafety cabinet to maintain aseptic conditions.
2. Cell Assessment Before Transfer
Cells are evaluated to ensure they are suitable for transfer.
Common checks include:
-
Visual inspection under a microscope
-
Confluency estimation
-
Detection of contamination
-
Assessment of cell morphology
Transferring unhealthy or contaminated cells compromises downstream experiments.
3. Detachment or Collection of Cells
Adherent cells may require enzymatic or mechanical detachment, while suspension cells are collected directly.
This stage is carefully controlled to avoid:
-
Excessive enzymatic exposure
-
Harsh mechanical agitation
-
Prolonged temperature changes
4. Cell Counting and Viability Measurement
Quantifying cells allows precise control of seeding density and experimental conditions.
Typical methods include:
-
Hemocytometer counting
-
Automated cell counters
-
Viability dyes
Accurate counts support consistency between experimental replicates.
5. Transfer to New Vessel or System
Cells are gently resuspended in fresh medium and transferred to the target container or system.
Important factors:
-
Proper volume and cell concentration
-
Gentle pipetting technique
-
Temperature-equilibrated media
6. Post-Transfer Incubation and Monitoring
After transfer, cells are placed in controlled conditions such as incubators with defined temperature, humidity, and gas composition.
Monitoring includes:
-
Visual inspection after attachment
-
Growth rate assessment
-
Morphological evaluation
Any abnormalities are documented and investigated.
Variables That Influence Transfer Success
Several technical and biological factors affect the outcome of a cell transfer protocol.
Key variables include:
-
Cell type and origin
-
Passage number
-
Media composition
-
Enzymatic reagents used
-
Temperature during handling
-
Duration of transfer process
Optimizing these parameters improves reproducibility and experimental reliability.
Sterility and Contamination Control
Contamination is one of the most common causes of failed cell experiments.
Preventive measures include:
-
Working in certified biosafety cabinets
-
Using sterile consumables
-
Regular equipment disinfection
-
Routine mycoplasma testing
-
Proper waste disposal
A robust cell transfer protocol explicitly addresses contamination risks.
Documentation and Traceability
Modern laboratories maintain detailed records for each transfer event.
Documentation typically includes:
-
Cell line identification
-
Passage number
-
Date and operator
-
Media and reagent batch numbers
-
Observations during transfer
This information supports quality control, regulatory compliance, and troubleshooting.
Automation in Cell Transfer Protocols
Automation is increasingly used to improve consistency and throughput.
Advantages of automated systems:
-
Reduced human error
-
Standardized handling
-
Higher reproducibility
-
Improved scalability
Robotic liquid handlers and closed-system transfer devices are particularly valuable in clinical-grade cell production.
Challenges in Implementing Cell Transfer Protocols
Despite standardization efforts, challenges remain.
Common issues include:
-
Variability between operators
-
Cell line-specific sensitivities
-
Equipment calibration errors
-
Environmental fluctuations
Ongoing training and protocol review help mitigate these risks.
Best Practices for Reliable Cell Transfers
Experienced laboratories follow several widely accepted practices:
-
Validate protocols for each cell type
-
Use gentle handling techniques
-
Maintain consistent timing
-
Avoid repeated temperature changes
-
Train staff thoroughly
-
Review protocols periodically
These steps significantly improve experimental consistency.
Choosing Reagents and Equipment
The quality of consumables directly affects protocol performance.
Selection criteria often include:
-
Compatibility with specific cell types
-
Sterility assurance
-
Supplier reliability
-
Regulatory compliance for clinical applications
Using standardized reagents simplifies protocol optimization.
Conclusion
The Cell Transfer Protocol is a foundational element of cell-based research and biomanufacturing. By defining how cells are moved, handled, and monitored, it safeguards cell health, ensures experimental reproducibility, and supports reliable scientific conclusions.
From basic laboratory research to advanced therapeutic development, investing in well-documented and carefully implemented transfer procedures reduces technical variability and strengthens data integrity. For any organization working with living cells, a robust protocol is not optional but essential.
Frequently Asked Questions
What is a cell transfer protocol used for?
A cell transfer protocol is used to safely and consistently move cells between containers, instruments, or experimental stages while preserving viability and preventing contamination.
How does a standardized cell transfer protocol improve research quality?
Standardization reduces variability between experiments, minimizes handling errors, and ensures that observed effects are biological rather than technical.
Can cell transfer protocols differ between cell types?
Yes. Different cell lines and primary cells have unique sensitivities, so protocols are often adapted to specific biological requirements while maintaining the same core principles.
What are the most common risks during cell transfer?
The most common risks include contamination, mechanical stress to cells, temperature shock, and incorrect cell concentration, all of which can compromise experimental outcomes.
Download PDF
Enter your email address to unlock the full PDF download.
Generating PDF...